Lamp size:


Fumes from cleaning principle: the use of efficient wavelength UV Germicidal Ultraviolet light irradiation lamp smoke, to change the molecular structure of oils and fats, so that polymer degradation into low molecular weight compounds, the effect is 3 times the normal 185nm. UV Ultraviolet Light using high-energy decomposition of oxygen molecules in the air of free oxygen, or reactive oxygen species, because of free oxygen by the imbalance in the processing of e+e portability and oxygen molecules, and produce ozone. Ozone has a strong oxidation of organic matter, and the smell of smoke has a strong role in the elimination. Using high-energy ultraviolet light and ozone molecular on the soot oxidation reaction photolysis, to convert water and carbon dioxide, and discharged through the exhaust pipe. UV Germicidal Lamps advantages : compared with standard germicidal lamp, produced under the same length, about 3 times the UV output of 13 thousand hours of long life, in a wide temperature range (4-40OC) to maintain stability high UV intensity with fewer lamps, lower initial installation and operating costs.

For Hot Cathode UV Germicidal Lamps
1. Radiate the cooking fume and change the molecular structure of the grease by the UV beam of the highly efficient Ultraviolet germicidal lamp and degrade the
high molecular compound to low one. The capability of our Hot Cathode UV germicidal lamp is 3 times than the normal germicidal lamp.
2. The UV beam of the highly efficient ultraviolet germicidal lamp can decompose the ozone molecules in the air and produce the free oxygen (active oxygen).
Since the free oxygen carries unbalanced electron-positron, it has to combine with oxygen molecules and in this case produces ozone. Ozone is extremely
oxidative to organic matter. So it can resolve the cooking fume and its off-flavour to H2O and CO2 which will be vented by the duct.
1-1 External quality
1-1-1 External quality is checked by sights or by a vernier caliper with precision of ±0.02mm
1-1-2 Glass tube should be transparent, clean, straight, without any fault of affecting UV rays to get through
1-1-3 Lamp caps should be fixed on the end of the lamp tightly and properly without any faults of affecting installment
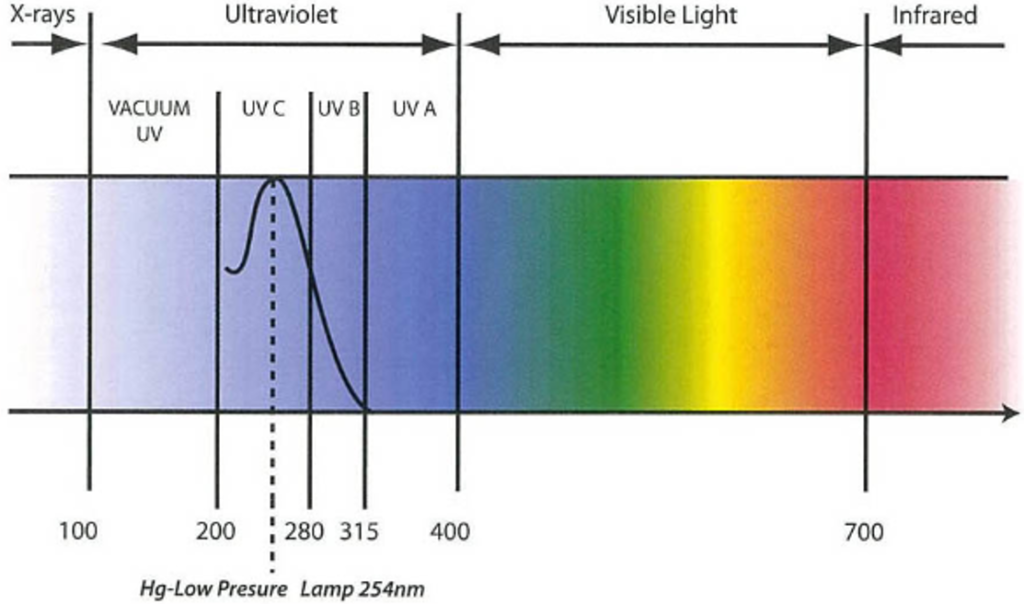
1-1-4 material for UV lamp tube should be quartz glass with can let 183.9nm and 253.7nm UV rays get through
1-2 List of Material
1. Appx 1
| No | Specification | Material | Remark |
|---|---|---|---|
| 1 | Glass tube | High-ozone quartz glass | |
| 2 | Filament | 98004 | |
| 3 | Exterior Wire | Molybdenum | |
| 4 | Sealed gas | Pure Argon | |
| 5 | Lamp cap | Ceramic | 2G11 |
| 6 | Mercury | Amalgam | 2 |
2-1 Operating temperature: 0℃ – 50℃
2-2 Operating humidity: 20% – 90% No dew and no icing
2-3 Preserving temperature: 0℃ – 70℃
2-4 Preserving humidity: <85% No dew and no icing
2. Appx 2
| No | Item | Code | Related Value |
Unit | Tolerance |
|---|---|---|---|---|---|
| 1 | Power of lamp tube | PL | 35.2 | W | |
| 2 | Voltage of lamp tube | UL | 74.6 | Vdc | |
| 3 | Electrical current of lamp tube | IL | 282 | mA | |
| 4 | 254(1) Strength of 254nm radiation (1M Distance) | UV | 28000 | vW/cm2 | |
| 5 | Steady time of lighting | Te | 10 | min | |
| 6 | Steady time of lighting | Te | 10 | min | |
| 7 | Average lifetime | H | 13000 | hours | |
| Testing Conditions 1. Ballast for testing 2. Instrument for testing: UI3000J electronic ballast synthetical performance testing instrument 3. Environment for testing: Temperature: 26±2℃ Moisture: 50% – 90% 4. (40601) Radio meter: UV-254 probe type(40601) |
|||||
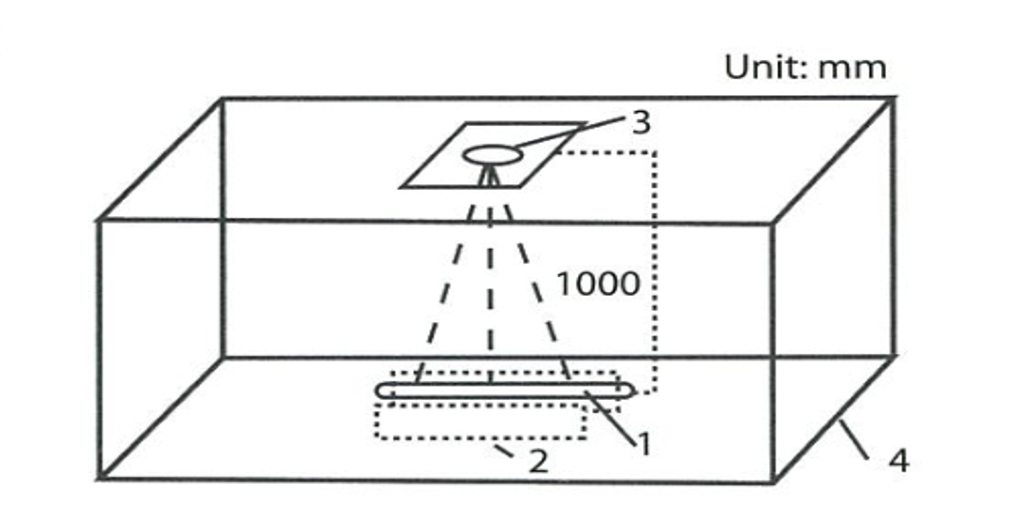
The test of measuring the strength of UV rays must be done in a black box. In testing, the lamp and the radiometer probe must be fixed on the position indicated in the beside chart precisely. The distance between the lamp and the probe should be flexible and be measured accurately.
(See chart A)
4-2 Testing Method
Fix the tested lamp on the table and light i t in accordance with its characters. Let the horizontal middle line of the lamp get through the center of the
radiometer probe. During lighting, the lighting surface (if any) should upright the lighting track and parallel with the receiving surface of the probe.
Install radiometer on the top of a black box and make it just illuminate the middle of the lamp. Let the normal of the receiving surface of the radiometer folds on the horizontal middle link of the tested lamp.
Adjust the position of the radiometer, let the receiving surface of the radiometer 1000+1 mm away from the surface of the testing map.
After the lamp being steady, open the quick door of the radiometer and get the reads form it directly.
Requirement of safe operation
During testing, operator should make some measure to prevent UV rays’ radiation from hurting their eyes and raked skin. It is recommended that this unit be installed and maintained by a trained technician.
Warming: Never look at UV lamps in operation. Unplug or disconnect power before re-lamping or servicing.
Vacuum testing of lamp tube is performed with electricity spark leak testing instrument.
6-1 Duration of guarantee is within 12 months after being sold
6-2 Preserving condition: In wrapping condition, temperature is 0℃ – 70
℃. Humidity is below 85% (No dew and no icing)
After testing, smoke mainly consist of: aldehydes, ketones, hydrocarbons, fatty acids, alcohols, aromatic compounds, esters, lactones. It also produced many types of harmful substances, such as: triethaolamine alias: 2.2”, 2”-trihydroxy triethylamine formula: N (CH2CH2OH) 3 the physical properties under room temperature are colorless and viscous liquid.
Rapid decomposition of harmful substances by ozone also known as
Trioxygen could eventually generate carbon dioxide(CO2), Water(H2O) and oxygen(O2). Smoke contains the following substances and how the ozone can help to decompose them by using the following chemical equation:
formaldehyde(formaldehyde): 3 HCHO + 2 O3 -> 3 H2O + 3 CO2
toluene(phenylmethane): C 6H5CH3 + 6 O3 -> 4 H2O + 7 CO2
xylene(xylene): C6H4(CH3)2 + 7 O3 > 5 H2O + 8 CO2
alcohol(ethanol): C2H5OH + 2 O3 -> 3 H2O + 2 CO2
propanol(propanol): 3 CH3COCH3 + 8 O3 -> 9 H2O + 9 CO2
CO(carbon monoxide): CO + O3 -> CO2 + O2
Phe P(Benzohyrene): C20H12 + 16 O3 -> 6 H2O + 20 CO2 + O2
Phe P(Benzphyrene) is a potent carcinogen, the most toxic of all